
Neither ILO nor WHO nor the European Commission shall be responsible for the interpretation and use of the information contained in this material. The published material is being distributed without warranty of any kind, either expressed or implied. Question Ask an American Elements Materials Science Engineer Chromium Sulfate Properties (Theoretical) Chromium Sulfate Health & Safety Information View and Print SDS About Chromium Sulfate Chromium Sulfate is a moderately water and acid soluble Chromium source for uses compatible with sulfates. The substance is toxic to aquatic organisms.Īll rights reserved. TLV: (as Cr(III), inhalable fraction): 0.003 mg/m 3, as TWA (DSEN) (RSEN) A4 (not classifiable as a human carcinogen). Repeated or prolonged contact may cause skin sensitization. Linear Formula: Cr2(SO4)3 xH2O CAS Number: 1 Molecular Weight: 392.18 (anhydrous basis) EC Number: 233-253-2 MDL number: MFCD00150215 PubChem Substance ID: 24861064 NACRES: NA.23 Recommended Products Sigma-Aldrich 8.02485 Chromium (III) acetylacetonate for synthesis View Price and Availability Sigma-Aldrich 8. The substance is irritating to the respiratory tract.Ī harmful concentration of airborne particles can be reached quickly when dispersed.Įffects of long-term or repeated exposure Chromium sulfate Cr2(SO4)3 or Cr2O12S0 - structure, chemical names, physical and chemical properties, classification, patents, literature. Substance name Solubility in other solvents Chromite Chromium(III) oxide Chromium(III) oxide, hydrated Chromium(III) sulfate, hydrated Chromium(III). With the financial assistance of the European Commission.ĭecomposes on heating. Prepared by an international group of experts on behalf of ILO and WHO, The pathway of the sparingly soluble salt can be easily monitored by x-rays. Its solubility product is 1.08 × 10 10 at 25C, so it is ideally suited for this purpose because of its low solubility when a barium milkshake is consumed by a patient. If appropriate, moisten first to prevent dusting. We mentioned that barium sulfate is used in medical imaging of the gastrointestinal tract. ammonium chromic sulphate, sulfuric acid, ammonium chromium(3+) salt. Sweep spilled substance into covered containers. Ammonium chromium(III) sulfate dodecahydrate. 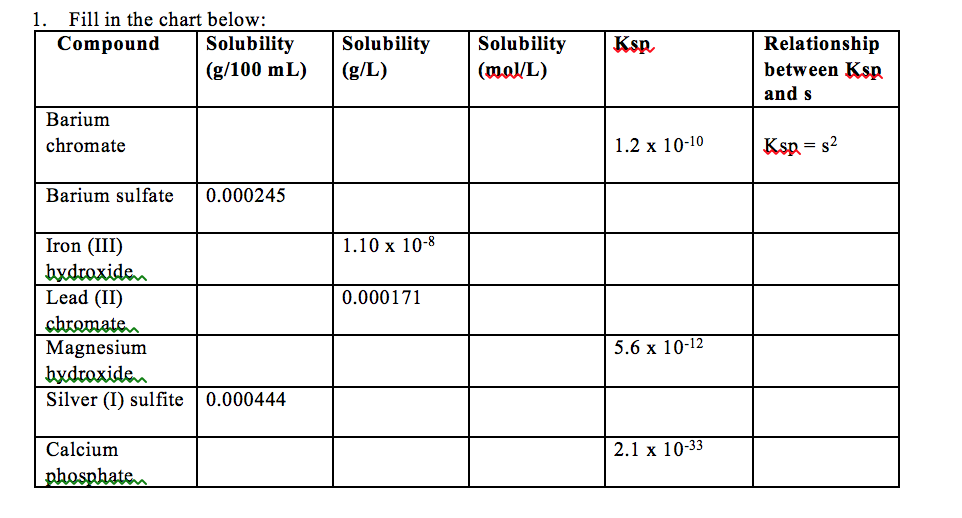
Solutions turn green when heated CHEMINFO Soluble in water Alfa Aesar. Solubility in Water: Decomposition: > 662F (350C). Chromium potassium sulfate dodecahydrate agent detailed information in. 
Vacuum spilled material with specialist equipment. This document provides a brief description of chromium hydroxide sulfate. Solubility: 250 g/l: Dimensions Materials Information Toxicological Information Safety Information according to GHS Hazard. Do NOT let this chemical enter the environment. Potassium chromium(III) sulfate, Alum chrome: Description: Chromium(III) potassium sulfate dodecahydrate: References Product Information CAS number: : EC number: 233-401-6: Grade: ACS,Reag. Personal protection: particulate filter respirator adapted to the airborne concentration of the substance. Chromium sulfate Cr2(SO4)3 or Cr2O12S0 - structure, chemical names, physical and chemical properties, classification, patents, literature, biological. Give one or two glasses of water to drink. Rinse skin with plenty of water or shower.įirst rinse with plenty of water for several minutes (remove contact lenses if easily possible), then refer for medical attention. Use local exhaust or breathing protection. Over the temperature range investigated, the decomposition pressures were significantly high so that chromium sulfate is not expected to form on commercial alloys containing chromium when exposed to gaseous environments containing oxygen and sulfur (such as those encountered in coal gasification).In case of fire in the surroundings, use appropriate extinguishing media. X-ray diffraction analysis indicated that the decomposition product was crystalline Cr 2O 3 and that the mutual solubility between Cr 2(SO 4) 3 and Cr 2O 3 was negligible. Over the temperature range studied, the change in the Gibbs’ free energy of formation of chromium sulfate Cr 2O 3(s) + 3SO 3(g) → Cr 2(SO 4) 3(s) can be expressed as ΔG 0 = Thermogravimetric and differential thermal analyses were used to follow the decomposition of chromium sulfate. The solid chromium sulfate was equilibrated in a gas stream of controlled SO 3 potential. These salts are usually either violet or green solids that are soluble in water. The stability of chromium (III) sulfate in the temperature range from 880 to 1040 K was determined by employing a dynamic gas-solid equilibration technique.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
